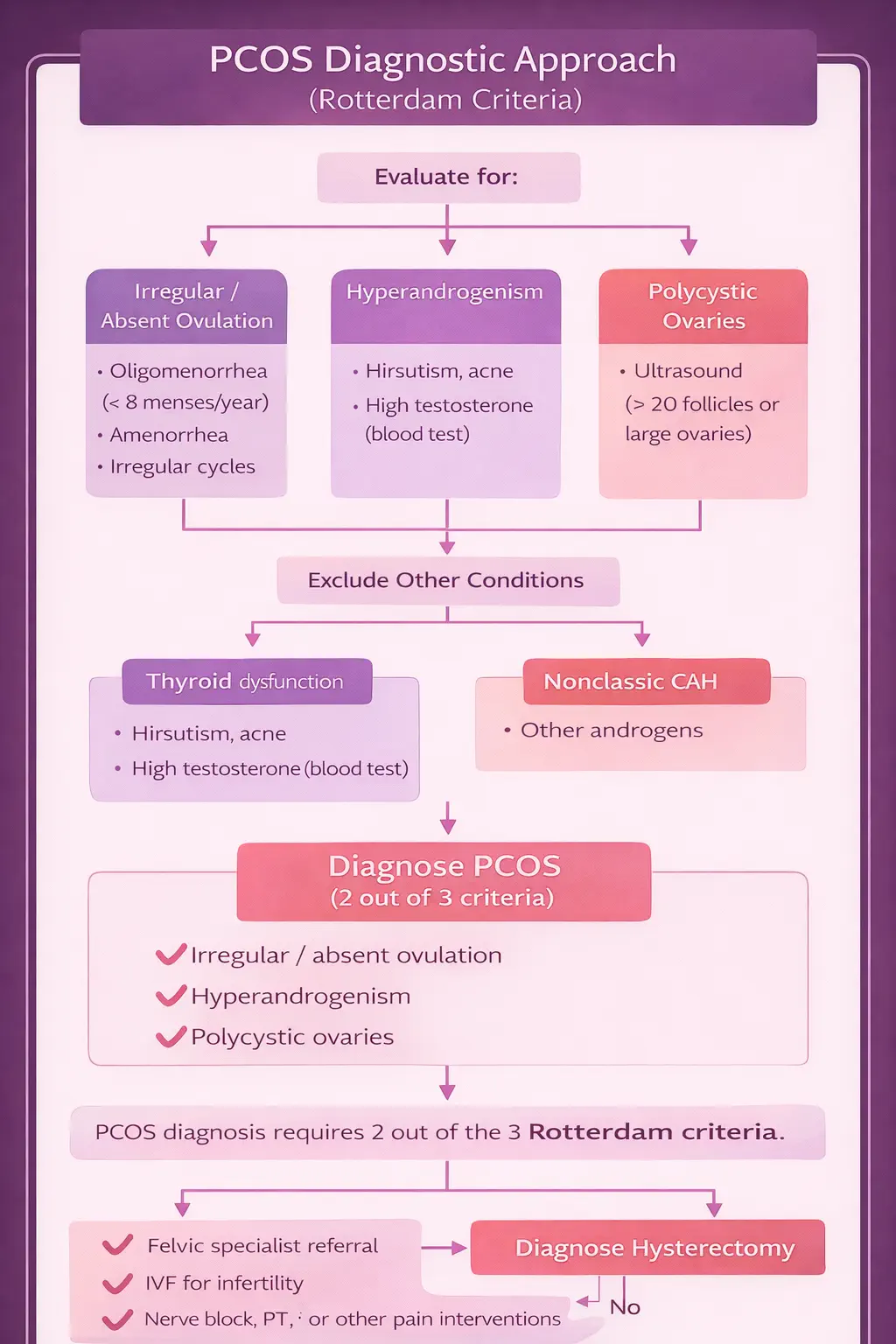
Because PCOS is a chronic condition with no cure, the goal of treatment
is to manage symptoms, restore hormonal balance, reduce metabolic risk, and address the
patient’s most pressing concerns — whether that is irregular periods, fertility,
acne, hirsutism, weight management, or long-term health. Treatment is individualized
and evolves over a woman’s lifetime as her goals change.
Lifestyle Modification
Lifestyle modification is the cornerstone of PCOS treatment regardless of
BMI. A balanced, anti-inflammatory diet, regular physical activity (both aerobic exercise and
resistance training), adequate sleep, and stress management all contribute to improved insulin
sensitivity, reduced androgens, and better overall outcomes. For women who are overweight,
even a 5–10% reduction in body weight has been shown to:
- Improve or restore ovulatory cycles
- Reduce testosterone and androgen levels
- Lower fasting insulin and improve insulin sensitivity
- Reduce the risk of progression to type 2 diabetes
- Improve mood, energy, and quality of life
Combined Oral Contraceptives (OCPs)
For women who are not currently trying to conceive, combined oral contraceptives
are a first-line treatment for menstrual irregularity and hyperandrogenism. OCPs work by:
- Regulating the menstrual cycle and providing regular withdrawal bleeds
- Suppressing ovarian androgen production
- Increasing sex hormone-binding globulin (SHBG), which binds free testosterone
- Protecting the endometrium from hyperplasia due to unopposed estrogen
OCPs containing an anti-androgenic progestin (such as drospirenone) may provide additional
benefit for acne and hirsutism. See our
birth control options page for more on
hormonal contraception.
Metformin
Metformin is an insulin-sensitizing medication originally developed for
type 2 diabetes. In PCOS, it targets the underlying insulin resistance that drives much of
the hormonal imbalance. Metformin can improve menstrual regularity, modestly reduce
androgen levels, and may facilitate weight loss. It is particularly useful in women with
PCOS who have documented insulin resistance, prediabetes, or type 2 diabetes. Metformin
can be used alone or in combination with OCPs.
Spironolactone
Spironolactone is an anti-androgen medication used specifically for the
cosmetic symptoms of PCOS — hirsutism, acne, and androgenic alopecia. It works by
blocking androgen receptors and reducing androgen production. Because spironolactone is
teratogenic (can cause birth defects), it must be used with reliable contraception and is
typically prescribed alongside an OCP. Full effect on hirsutism takes 6–12 months.
Inositol
Myo-inositol and D-chiro-inositol are nutritional
supplements with emerging evidence supporting their use in PCOS. They act as insulin
sensitizers and may improve ovulation rates, hormonal profiles, and metabolic parameters.
The 2023 international evidence-based guidelines acknowledge inositol as a potential
adjunctive therapy, though they note that larger, higher-quality trials are still needed.
Many patients find inositol helpful as a complement to standard medical therapy, and it
is generally well-tolerated.
Women with PCOS who are not on hormonal contraception and have irregular periods
are at increased risk for endometrial hyperplasia and endometrial cancer. When
ovulation does not occur, the uterine lining is exposed to estrogen without the protective
effect of progesterone (unopposed estrogen). Over time, this can lead to abnormal thickening
of the endometrium. If you have PCOS and go more than 3 months without a period while not
on contraception, contact your provider for evaluation. A progestin withdrawal or endometrial
assessment may be necessary.