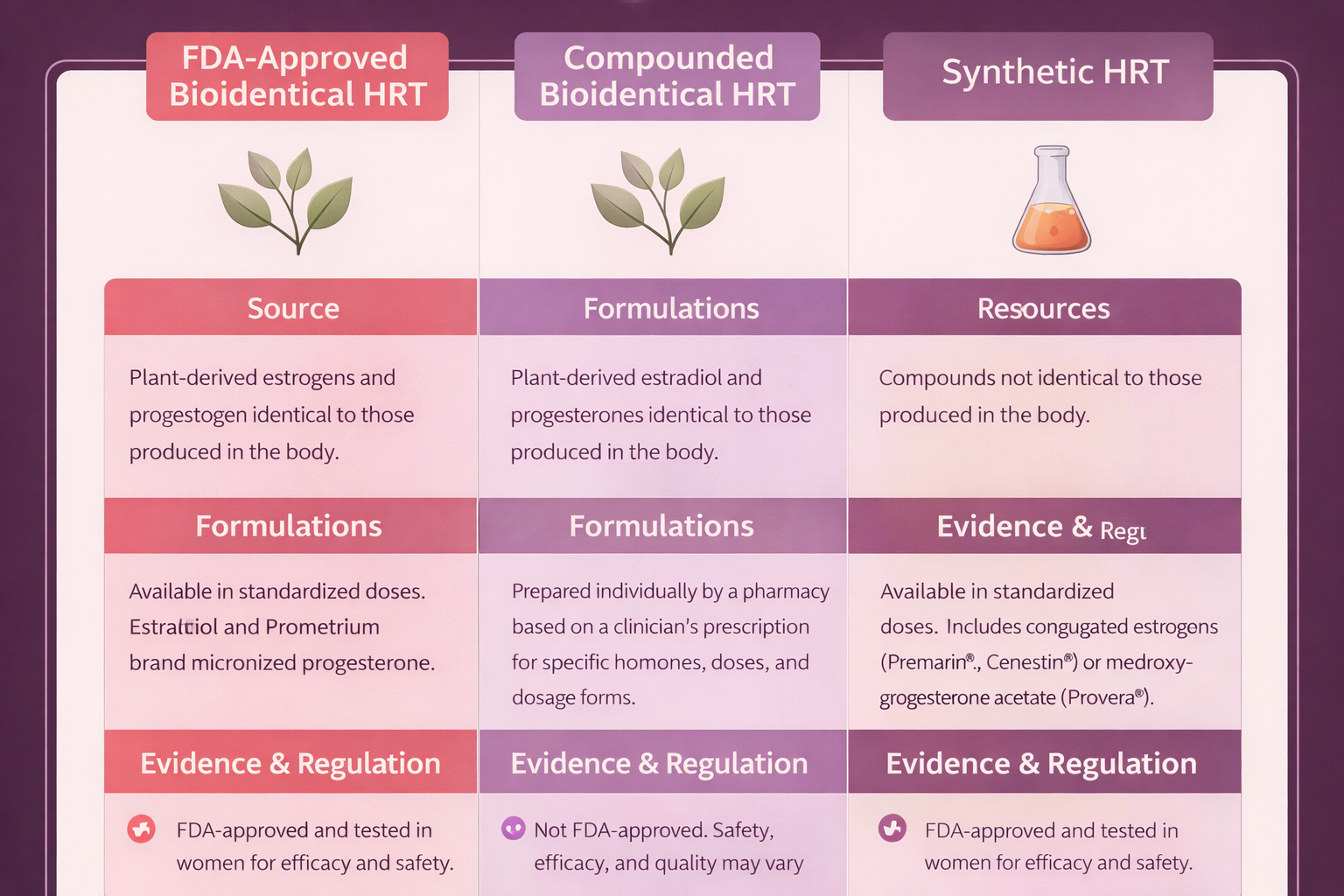
| FDA Regulation |
Yes — full pre-market review |
No — pharmacy-level oversight only |
Yes — full pre-market review |
| Potency Consistency |
Standardized; batch-to-batch uniformity required by cGMP |
Variable; studies document 67–150% of labeled dose |
Standardized; batch-to-batch uniformity required by cGMP |
| Purity Testing |
Required — contaminant limits enforced |
Pharmacy-dependent; not standardized |
Required — contaminant limits enforced |
| Safety Data |
Robust — clinical trial data required for approval; post-market surveillance |
Limited — no pre-market clinical trials; inferred from FDA-approved bioidentical data |
Extensive — WHI and decades of additional trial data |
| Efficacy Data |
Demonstrated in clinical trials |
Not independently demonstrated; assumed from bioidentical molecule data |
Demonstrated in clinical trials |
| Molecular Structure |
Identical to human hormones (17β-estradiol, progesterone) |
Identical to human hormones (same molecules, different manufacturing) |
Different from human hormones (equine estrogens, synthetic progestins) |
| Typical Cost |
Moderate — many covered by insurance; generics available |
Variable — often not covered by insurance; may be higher out-of-pocket |
Low to moderate — generics widely available; typically covered |
| Customization |
Fixed doses and delivery systems; multiple options available |
Flexible dosing and combinations; custom formulations possible |
Fixed doses; fewer delivery options than bioidentical estradiol |
| Patient Labeling |
Standardized — FDA-approved package inserts with warnings, contraindications, interactions |
Not standardized — no required patient information insert |
Standardized — FDA-approved package inserts with warnings, contraindications, interactions |
| ACOG/Endocrine Society Position |
Recommended when bioidentical is preferred |
Recommended only when FDA-approved equivalent unavailable |
Appropriate for many patients; extensive evidence base |