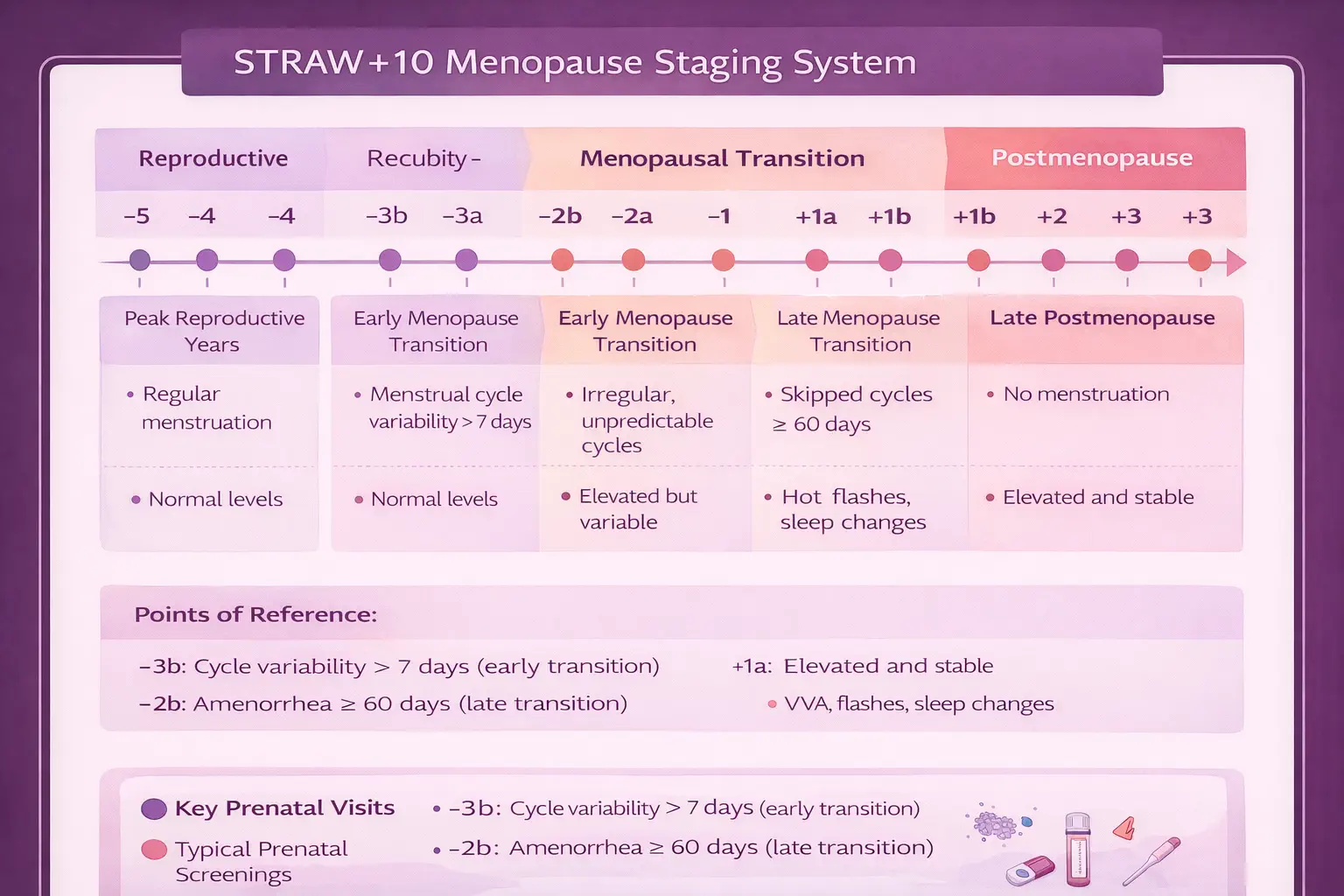
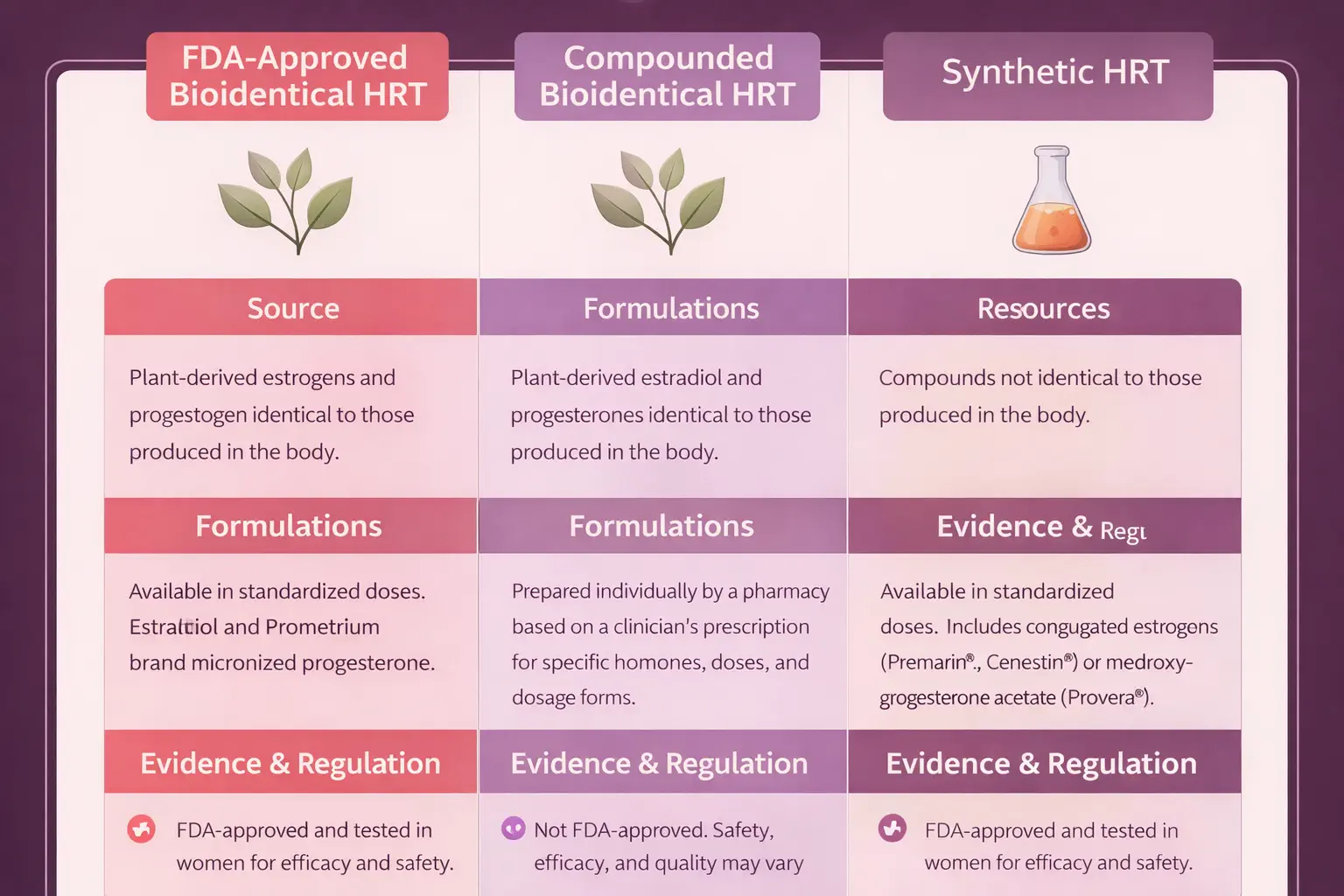
| Baseline (Pre-HRT) |
Comprehensive history, symptom assessment, cardiovascular risk evaluation, breast cancer risk assessment (Tyrer-Cuzick or similar), mammogram, BMD (if indicated), lipid panel, liver function, blood pressure |
Establish candidacy, identify contraindications, document baseline risk |
| 3-Month Follow-Up |
Symptom response evaluation, side effect assessment, blood pressure, dose adequacy review |
Confirm therapy is effective, adjust dose/route if needed, address early side effects |
| Annual Review |
Symptom reassessment, mammogram, BMD (per USPSTF schedule), lipid panel, blood pressure, breast exam, pelvic exam, reassessment of ongoing candidacy |
Verify continued appropriateness, screen for new contraindications, evaluate benefit vs. risk |
| As Needed |
Unscheduled bleeding evaluation (endometrial biopsy or transvaginal ultrasound), new symptom assessment |
Rule out endometrial pathology, address emerging concerns |