Medical management is the first-line approach for endometriosis-related
pain in most patients. The goals are to suppress the hormonal stimulation
that drives endometriotic tissue growth, reduce inflammation, and manage pain. It is
important to understand upfront: medical management suppresses endometriosis
but does not cure it. Symptoms typically recur when treatment is discontinued,
because the underlying implants remain in place.
Pain Management: NSAIDs
Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and naproxen are
the foundation of symptomatic pain relief. They work by inhibiting prostaglandin
synthesis, which drives both inflammation and uterine cramping. NSAIDs are most
effective when taken before the onset of pain (i.e., starting 1–2
days before expected menstruation) rather than waiting for pain to become established.
They are typically used alongside hormonal suppression, not as standalone therapy
for confirmed endometriosis.
Hormonal Suppression Options
The principle behind hormonal therapy for endometriosis is straightforward:
endometriotic tissue is estrogen-dependent. Reducing estrogen stimulation reduces
the activity and growth of implants. Several hormonal approaches are available,
and the choice depends on the patient’s symptoms, fertility plans, side
effect tolerance, and response to prior treatment.
Combined oral contraceptives (COCs) are often the first hormonal
agent tried, particularly in younger patients or those who also need contraception.
Continuous use (skipping the placebo week to avoid withdrawal bleeding)
is preferred over cyclic use for endometriosis management, as it provides more
consistent suppression and eliminates the menstrual flare that many patients experience.
Progestins provide effective suppression for many patients.
Options include norethindrone acetate (oral, 5 mg daily), dienogest
(2 mg daily — specifically studied and approved for endometriosis in
many countries), the levonorgestrel intrauterine device (Mirena), and
medroxyprogesterone acetate (Depo-Provera). Progestins work by decidualizing and
atrophying endometriotic tissue, reducing inflammation, and often reducing or
eliminating menstruation.
GnRH agonists such as leuprolide (Lupron) create a temporary
menopausal state by suppressing the hypothalamic-pituitary-ovarian axis after an
initial “flare” phase. They are highly effective for pain control but
cause significant menopausal side effects (hot flashes, vaginal dryness, bone
density loss). For this reason, GnRH agonists are used with add-back
therapy — low-dose norethindrone with or without conjugated estrogen
— to mitigate side effects while maintaining therapeutic benefit. Use is
generally limited to 6 to 12 months due to the risk of bone
density reduction.
GnRH antagonists represent a newer class. Elagolix
(Orilissa) is FDA-approved specifically for the management of endometriosis
pain. Unlike GnRH agonists, elagolix is an oral medication that provides dose-dependent
estrogen suppression without the initial flare phase. At lower doses (150 mg
once daily), it produces partial estrogen suppression and can be used for up to
24 months; at higher doses (200 mg twice daily, with add-back), the treatment
duration is limited to 6 months. Elagolix offers a more nuanced approach to estrogen
management than the all-or-nothing suppression of GnRH agonists.
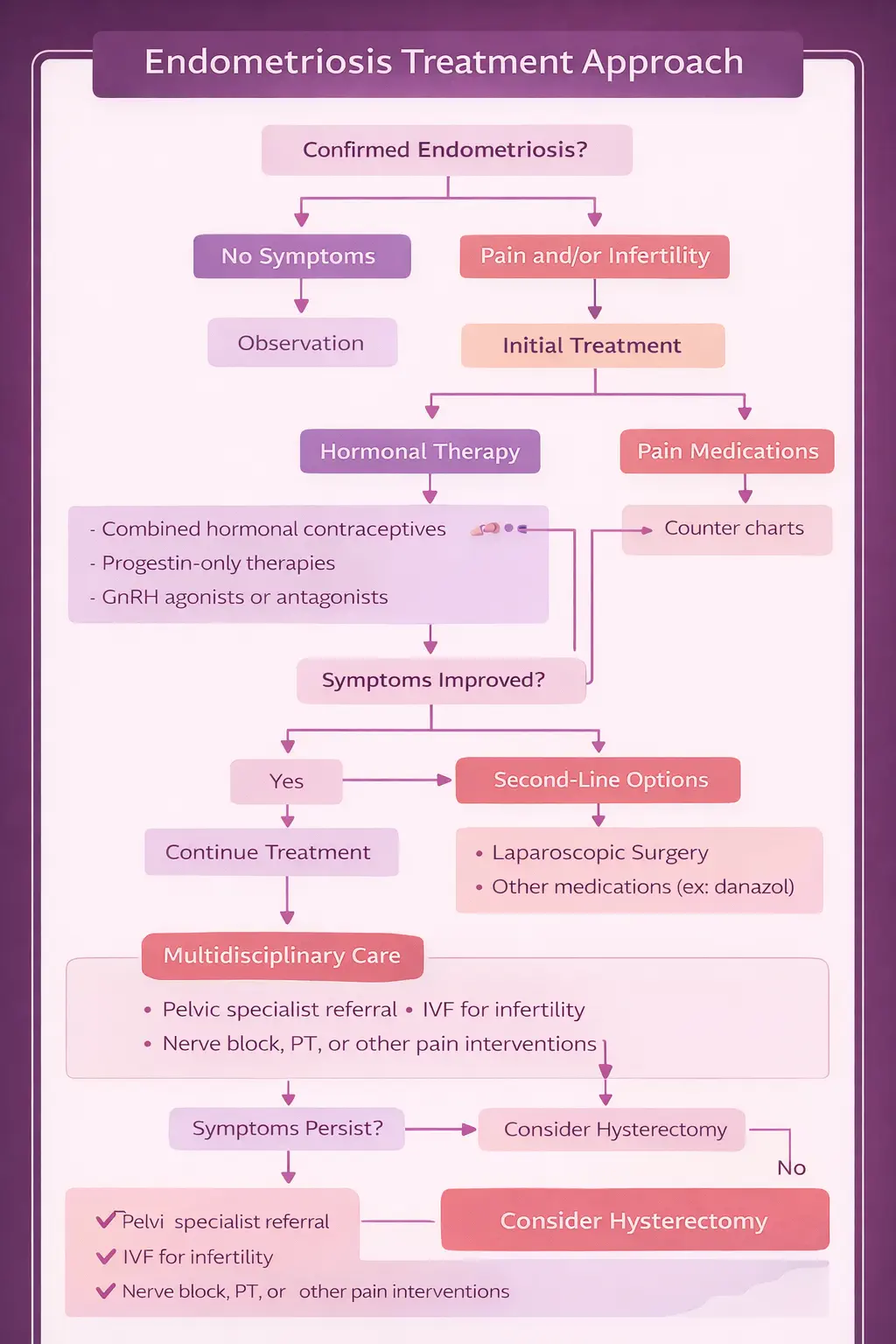
ACOG Practice Bulletin #114 recommends empiric treatment with hormonal therapy for
women with clinically suspected endometriosis without requiring surgical
confirmation first. If a patient has characteristic symptoms and responds
to hormonal suppression, this supports the clinical diagnosis and surgery can be
deferred. Surgery is pursued when medical management fails, when histologic
diagnosis is needed, or when fertility optimization requires it.